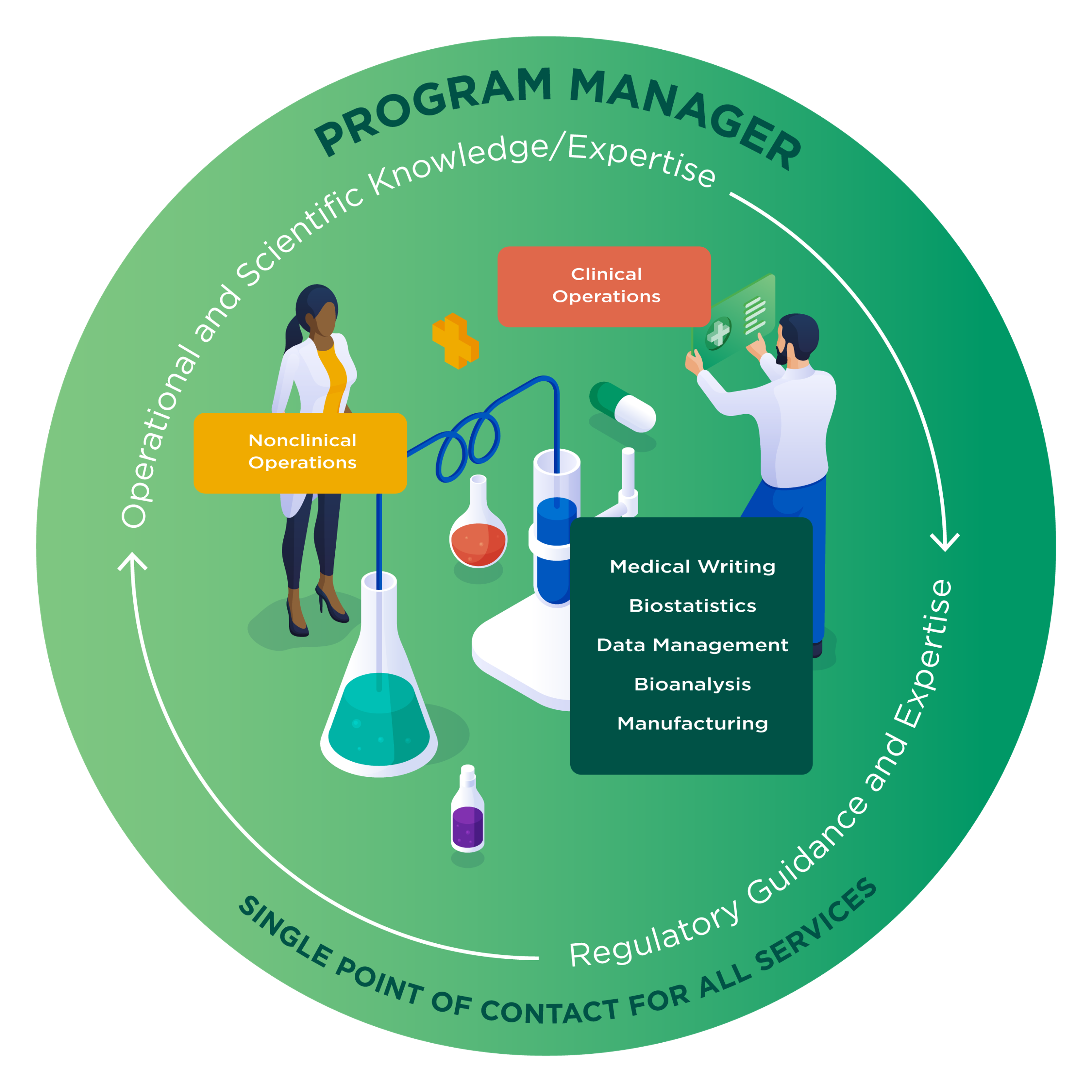
Program Management for One Point of Contact
With Altasciences, a dedicated cross-functional program manager leads your development program from start to finish.
Our integrated team advances your molecule from preclinical testing through early-phase clinical studies using a proactive, project-specific approach that unites bioanalytical services, preclinical safety evaluations, formulation development, clinic-ready manufacturing, and clinical testing through proof of concept.
With centralized oversight and seamless collaboration across sites, our scientific and operational teams work efficiently, conducting activities in parallel whenever possible to reduce timelines and costs.
Access our fact sheet for more information on our unique Program Management offering.End-to-End Drug Development: Simplified and Efficient
Your dedicated, cross-functional program manager:
- Provides you with a centralized point of contact to improve speed, efficiency and communication.
- Manages your study timelines proactively using our proprietary scheduling system.
- Leverages our team of experts to review your emerging data and make strategic study design or timeline adjustments while continuously supporting your needs.
- Responds to your program challenges in real time.
- Shares all your information across departments proactively, so you only have to Tell Us Once™.
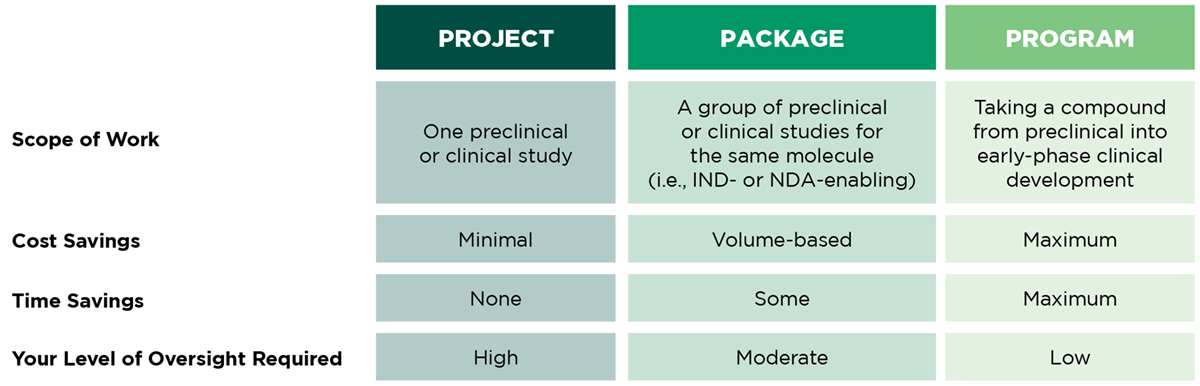
Shifting to a Program Model
Our program management model enables an optimized drug development experience with a single CRO/CDMO, increasing efficiencies for your entire program.